
With fewer 90° LP–BP repulsions, we can predict that the structure with the lone pair of electrons in the equatorial position is more stable than the one with the lone pair in the axial position. It has a molecular geometry of the formula AX4E it forms a see-saw shape and has a trigonal bipyramidal molecular geometry. 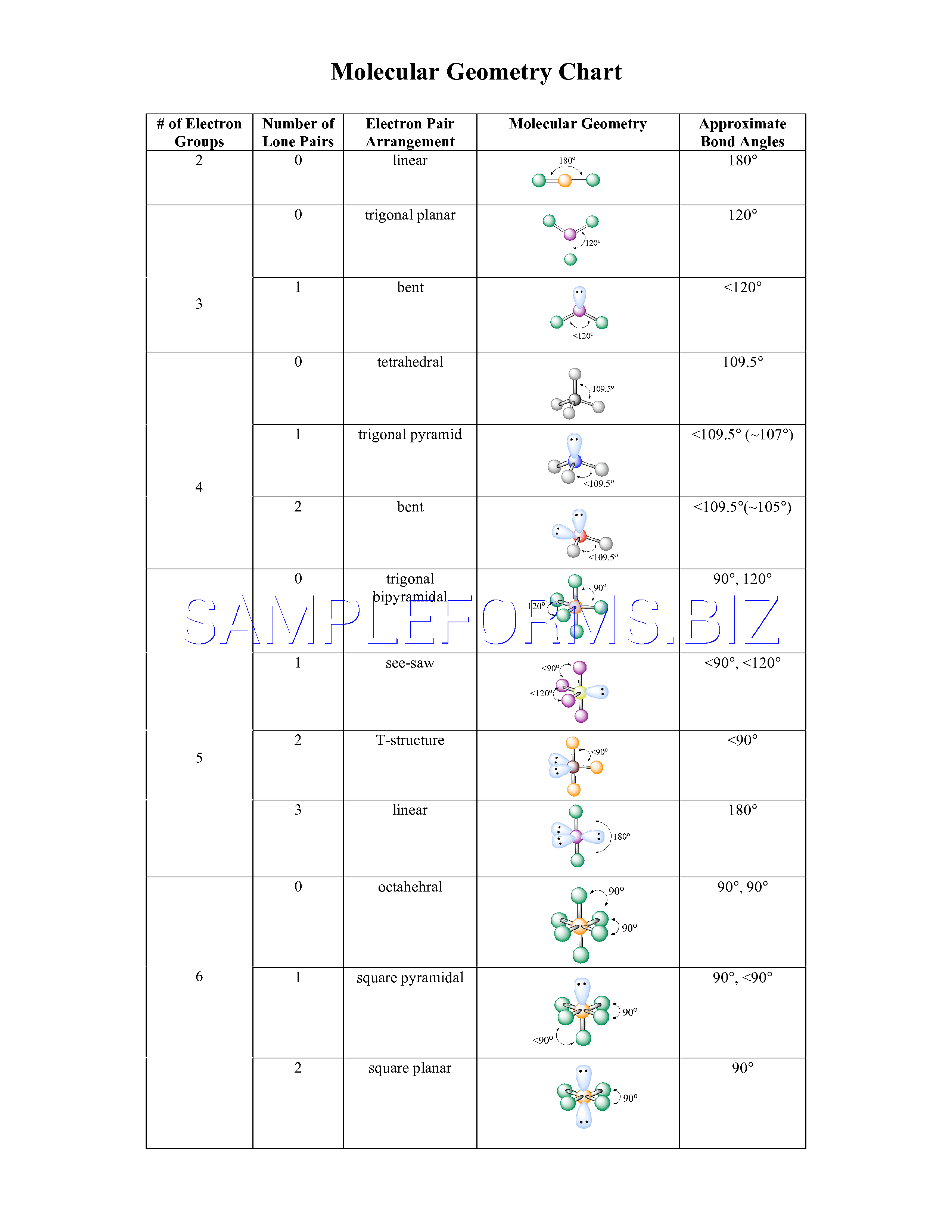
There are three lone pairs on each fluorine atom. If we place it in the axial position, we have two 90° LP–BP repulsions at 90°. Sulfur Tetrafluoride has 34 valence electrons, out of which it forms four covalent bonds and one lone pair of electrons on the central atom in its Lewis structure. However, because the axial and equatorial positions are not chemically equivalent, where do we place the lone pair? If we place the lone pair in the equatorial position, we have three LP–BP repulsions at 90°. Formal charge (FC) Valence electrons 0. Lewis structures also called Lewis dot formulas, Lewis dot structures, electron dot structures, or Lewis electron dot structures (LEDs) are diagrams that show the bonding between atoms of a molecule, as well as the lone pairs of electrons that may exist in the molecule. The formula for the formal charge is as follows. Let us calculate the formal charges on each of the constituent atoms. The Lewis structure of IF3 shows that I is surrounded by 2 lone pairs of electrons and forms 3 single bonds with each of the F. As you already know, valence electrons are used to draw the Lewis structures of any molecule. The molecule is neutral, i.e., there is no charge on it. Before we begin to learn the steps for drawing Lewis structure let’s see the Lewis structure of IF3. We designate SF 4 as AX 4E it has a total of five electron pairs. Carbon will be singly bonded to H, H, Cl, and Cl, as shown in the Lewis structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
