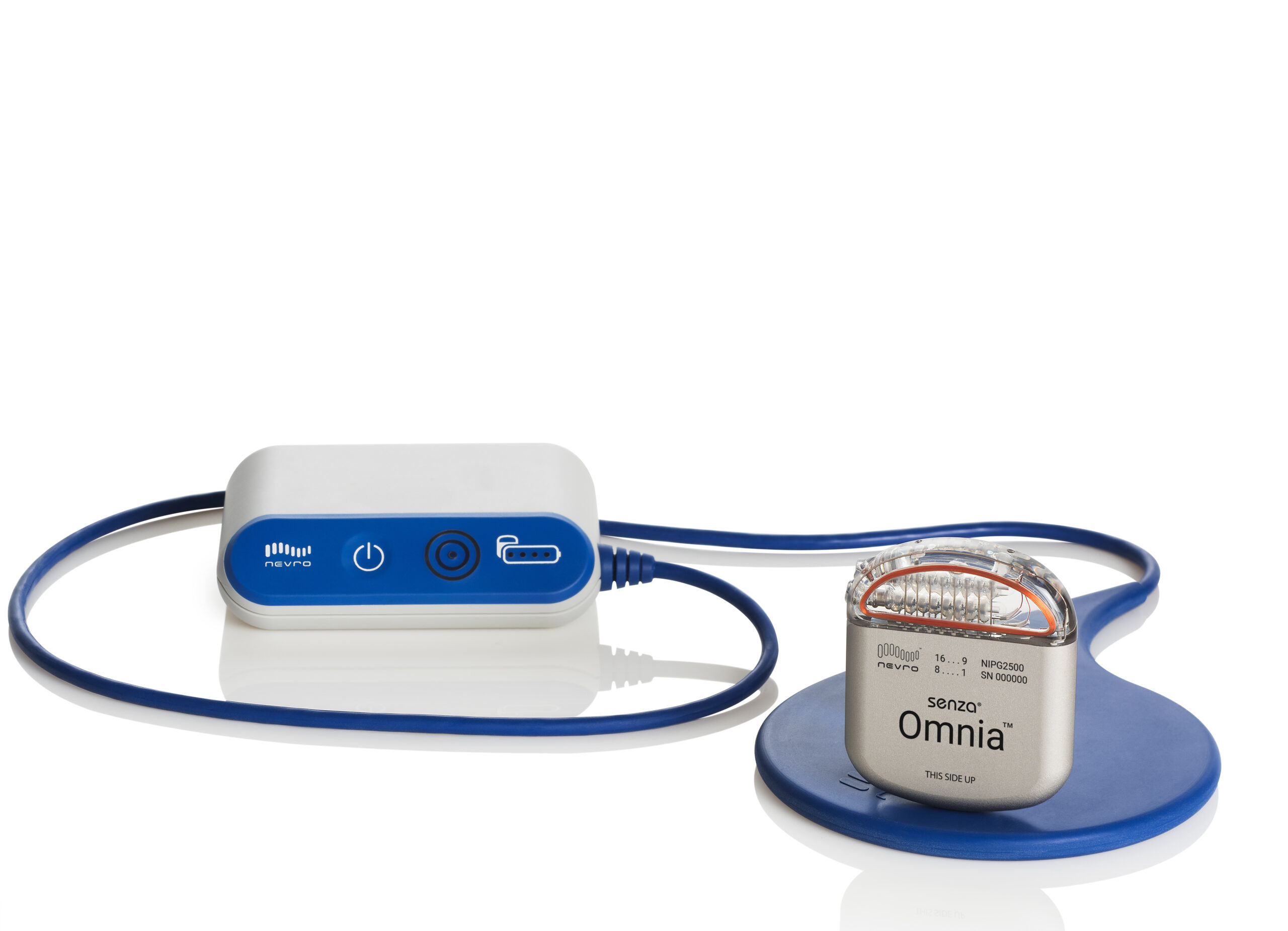
portfolio of SCS technology, which includes the Proclaim SCS family and the Eterna SCS platform. In a multicenter study of BurstDR spinal cord stimulation, 100 of participants on a low-energy programming settings felt full pain relief on less than six hours of battery use per day, and 43 of participants achieved pain relief on less than 2 hours of battery use per day. The company said this new indication will apply to its entire U.S. “This new indication for Abbott’s SCS devices, together with BurstDR stimulation, allows physicians the ability to identify and treat a new group of people, providing them with relief from chronic back pain.” 4, 2022 / PRNewswire / - Abbott (NYSE: ABT) announced today that the U.S. “To date, we have struggled with how to treat people who weren’t considered a good surgical candidate because we didn’t have clear, data-driven treatment options for nonsurgical back pain,” Timothy Deer, MD, FIPP, DABPM, president and CEO of the Spine and Nerve Centers of the Virginias in Charleston, West Virginia, said in the release. This technology has been shown to improve ability to perform daily activities and reduce emotional suffering associated with chronic pain. The device may be unable to exit MRI mode and resume therapy. For people living with chronic pain and movement disorders, our comprehensive portfolio of therapies helps them move and feel better, by delivering stimulation to the spinal cord, dorsal root ganglion (DRG), or brain, depending on the needs of each patient. “This FDA expanded indication approval for our SCS devices is a significant step forward in Abbott’s goal to provide treatment access to those who suffer daily with chronic back pain but are not eligible for corrective surgery,” Pedro Malha, Abbott’s vice president of neuromodulation, said in the release.īurstDR uses pulses of mild electrical energy without paresthesia to change pain signals as they travel from the spinal cord to the brain, the company explained in the release. The Abbott Proclaim and Infinity neurostimulation systems deliver low-intensity electrical impulses to nerve structures. 
In August 2022, the FDA granted premarket approval for Abbott’s Proclaim Plus SCS system featuring FlexBurst360 therapy, the next generation of Abbott’s BurstDR stimulation technology, for chronic pain across up to six areas of the body. The approval was based on results from the DISTINCT study, which showed that patients with chronic back pain who were treated with the BurstDR spinal cord stimulation (SCS) technology reported improvements in pain levels, ability to perform daily activities and emotional well-being, according to an Abbott press release. When we experience an injury or physical trauma, nerve endings known as pain receptors send a signal along the spinal cord to the brain. Neurostimulation interrupts pain signals. If you continue to have this issue please contact to HealioĪbbott has announced the FDA expanded the indication of its BurstDR spinal cord stimulation devices for treating chronic back pain in those who have not had or are ineligible for back surgery. Neurostimulation, also known as spinal cord stimulation (SCS) therapy is a well-established pain treatment used by doctors for more than 55 years.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
